Animal studies:
Annual report 2024
The University of Groningen (UG) and the University Medical Center Groningen (UMCG) conduct animal experiments within the scope of research and teaching, because some important and relevant questions cannot be answered without the use of laboratory animals.
We are open about these activities and have created this website to show how we conduct animal studies and what we take into consideration in such testing. This is our contribution to the social debate about animal experiments in which anyone can form a considered opinion.
Dutch Transparency Agreement on Animal Testing
The importance of good and transparent communication about animal research is advocated by an increasing number of people. The University of Groningen is one of 15 organizations in the Netherlands that have signed the Netherlands Transparency Agreement on animal research. The aim of this Transparency Agreement is to create a more open and transparent climate around animal research. The signatories comprise universities, university medical centres, scientific institutes, companies, and associations that are all involved in animal research.
The signatories are Amsterdam UMC, Biomedical Primate Research Centre, Charles River Laboratories Den Bosch B.V., Envigo RMS B.V., Erasmus University Medical Center, the Royal Netherlands Academy of Arts and Sciences, Leiden University, the Netherlands Cancer Institute, Radboudumc, Radboud University, the University of Groningen, Maastricht University, Sportvisserij Nederland (the Dutch Angling Association), VU Amsterdam, Wageningen University & Research, Hubrecht Institute, Netherlands Institute for Neuroscience, Netherlands Institute of Ecology, TNO, LUMC, Noldus Information Technology, UMCG and University of Amsterdam.
Transparency
It is determined by law that Dutch research institutes provide information about the animal research that they carry out; however, this information is not always accessible and understandable for everyone. The signatories hope that this agreement will make a positive contribution to the openness around animal research.
Commitments
The signatories are involved in carrying out, supporting, or funding animal experiments for the benefit of human and animal health, quality of life, and nature and the environment. By signing this agreement, the organizations have made the following four commitments:
- We are clear about when, how, and why we use animals in research.
- We aim to improve the communication about animal research in the Netherlands with the public and with the media.
- We will be proactive in providing opportunities for the public to inform themselves about animal research and the regulations that govern it.
- We will report on our progress annually and share our experiences.
This agreement has been drawn up by Dutch researchers in collaboration with the European Animal Research Association (EARA) and Stichting Informatie Dierproeven (SID, the Animal Research Information Foundation). The Netherlands Transparency Agreement on animal research is based on existing agreements that have been drawn up in Belgium, Germany, France, Portugal, Spain, and the United Kingdom.
In 2024, we continued to uphold the core principles of the Transparency Agreement signed by the University of Groningen (UG) and the UMCG. Over the past year, we organized 53 guided tours for more than 400 participants in the animal facility of the UMCG. These visitors came from a wide range of backgrounds — including UMCG staff interested in learning more about the UMCG animal facility, as well as secondary school students, MBO students, and colleagues from other animal facilities.
Animal testing statistics
At the University of Groningen, animal procedures are conducted for fundamental and translational (applied) research, as well as for educational purposes. These procedures take place in the facilities at the UMCG (CDP) and FSE (FDD), as well as in the field.
In 2024, a total of 10,553 animal procedures were carried out, primarily involving mice, rats, birds, and fish. In 2022 and 2023, more than 14,000 procedures were conducted. The year 2024 marked the largest decrease in the number of animal procedures since 2017 — a drop of nearly 30%, and almost 45% since 2019 (the year with the highest number of procedures in the past eight years).
The figure below provides an overview of the total number of procedures per species over the last five years, showing trends such as a gradual decline in the use of most species. The number of animal procedures fluctuates annually due to variations in available funding and research capacity.
In 2024, the number of animal experiments at the University of Groningen (UG) further decreased. This decline is especially visible in the use of rats, which dropped by approximately 30% compared to previous years. Mice remain the most commonly used species, but the number of experiments involving mice also shows a downward trend. The number of experiments involving zebrafish was limited; much of the research is conducted on larvae younger than five days old. These are not counted as animal experiments under current regulations but still yield important scientific results. The number of experiments involving other fish species remained comparable to the previous year.
This decline aligns with both local and national efforts to develop alternatives to animal experimentation. Nevertheless, experiments sometimes remain necessary to answer complex research questions for which no alternatives are available. In December 2024, over 400 professors and administrators—including staff from the UG and the UMCG—signed a letter emphasizing the importance of continuing to conduct animal experiments when no alternatives are available.
A key point of attention is the use of both male and female animals in research.
This prevents breeding surpluses and ensures that scientific findings are applicable to the entire population. In 2024, 39% of the animals used were male and 29% were female. For 32% of the animals, the sex could not be determined—typically due to the use of larval animals or species that do not have distinguishable sexes. Assuming half of these animals were male, the male-to-female ratio of experimental animals at the UG in 2024 would have been 55.4% male and 44.6% female.
Number ofanimal experiments by sex
| Animals (all species) | 2024 | 2024 (%) |
| Males | 4,161 | 39% |
| Females | 3,019 | 29% |
| Unknown | 3,373 | 32% |
| Total | 10,553 | 100% |
The RUG is committed to promoting balanced representation of sexes in animal research and will continue to monitor and report this ratio in future annual reports.
Why animal experiments?
Staying healthy while getting older (Healthy Ageing), adapting to changing circumstances (Adaptive Life) and creating a robust society (Sustainable Society) are policy spearheads of the UMCG and the UG. Many of our research programmes therefore focus on issues such as healthy ageing, Alzheimer’s disease, diabetes and Parkinson’s disease, which sometimes require animal testing. Animal experiments are also required to study ecological phenomena such as bird migration.
Animal experiments at the UG/UMCG
The University and the UMCG want their fundamental and applied research programmes to be among the best in the world. We wish to conduct the animal studies required to achieve this goal in the best possible manner, which means providing optimal care to lab animals and safeguarding of their welfare as well as optimal facilitation of the animal experimenters.
Our animal tests are conducted at the UMCG (53%) and the Faculty of Science and Engineering (FSE, 47%), where animal testing is concentrated in several research institutes.
▶ Behavioural & Physiological Ecology Group
Research into the behaviour of animals in their natural surroundings
▶ Conservation Ecology Group
Research into the impact of habitat changes on organisms
▶ Theoretical Research in Evolutionary Life Sciences (TRES)
Focus on theorical developments in evolutionary ecology, behavioural sciences and evolutionary systems biology
▶ Evolutionary Genetics, Development & Behaviour (EGDB)
Research into the proximate cause of phenotypic diversity and its ecological and evolutionary consequences
▶ Genomics Research in Ecology & Evolution in Nature (GREEN)
Research into ecological, evolutionary and conservationist issues in relation to biodiversity, ecosystems, living environment interactions, speciation, adaptation and plasticity
▶ Neurobiology
Research into the role of the brain in the capacity of animals and humans to adapt to challenges and opportunities in the environment
▶ Groningen Research Institute of Pharmacy (GRIP)
Fundamental and applied pharmaceutical research
▶ Groningen University Institute for Drug Exploration (GUIDE)
Development of new medication
▶ Health Research and Epidemiology (SHARE)
Fundamental and applied research into factors that help people to stay healthy while getting old (Healthy Ageing)
▶ European Research Insitute for the Biology of Ageing (ERIBA)
Fundamental research into factors causing ageing
▶ Biomaterials (W.J.Kolff Institute)
Applied research into biomaterials and implants
▶ Fundamental, Clinical and Translational Cancer Research (Cancer Research Center Groningen)
Fundamental and applied research into oncology and tumour development
The research examples in this annual report illustrate research at the UG and the UMCG. An overview of the departments that conduct animal experimental research is available on the UG website.
The University of Groningen and the UMCG are in dialogue with various stakeholders in the field of animal testing. Its purpose is to both share and exchange information. For example, the dialogue is used to properly explain the value of animal experiments performed by the University of Groningen and UMCG to society and science. On the other hand, it offers the possibility to receive signals about animal testing and to follow up within the organization.
Legislation and regulations
Animal studies are governed by strict legislation and regulations. Since 1977, the welfare of lab animals used in the Netherlands has been protected by the Wet op de dierproeven (Wod). To supplement this act, the Dierproevenbesluit (Animal Experiments Decree) was adopted in 1985. The principle underlying the act is the ‘No, unless’ principle: animal experiments are only allowed if there are no alternatives. If researchers can conduct a study by using a computer model or slaughterhouse material, for example, they will not be allowed to use animals for their experiments.
With the Wod, the Netherlands had good legislation governing the use of lab animals. There were major differences with other countries, however, including European member states. To achieve identical legislative standards – at least within the European Union – guidelines were drafted, which in the Netherlands led to a revision of the Wod in 2015. The current Wod defines an animal experiment as ‘any use, invasive or non-invasive, of an animal for experimental or other scientific purposes, with known or unknown outcome, or teaching purposes, which may cause the animal a level of pain, suffering, anxiety or lasting harm equivalent to, or higher than, that caused by the introduction of a needle according to good veterinary practice’. Experiments conducted on animals without an endoskeleton, such as worms, snails and insects, are not covered by the Wod. The intention of the Wod is to protect lab animals in the Netherlands. One of its clauses stipulates, for example, that only qualified personnel are allowed to use lab animals and only within institutions that have a permit for such use.
In the old Experiments on Animals Act, two definitions were used for research involving wild animals: one covering their use in the laboratory and one covering animals living in nature. This distinction is no longer made in the newWod, which means that the same definition covers all animal testing, including research involving wild animals, whether in the laboratory or in their biotopes. It soon appeared that research involving wild animals in their biotopes was not covered in sufficient depth in the memorandum with the title ‘Wanneer is er sprake van een dierproef in de zin van de wet?’ (‘When is an experiment an animal experiment under the Act?’, in Dutch only) which was published on theCCDwebsite on 3 October 2016. A project group was therefore established with representatives from the relevant fields. In collaboration with theCCDand theNVWA, in 2017 the group published guidelines with the title ‘Dierproeven met wilde dieren in hun biotoop’ (‘Animal experiments with wild animals in their biotopes’, in Dutch only). UG researchers were involved in the formulation of these guidelines, which are used by UG researchers applying for and implementing animal experiments in nature.

Codes of Practice
Although legislation provides frameworks, it does not concern itself with details. For this reason, its specific interpretation may be unclear. Experts have therefore drafted several Codes of Practice covering various research fields: ‘Animal experiments in Cancer Research’ (1999), ‘Immunization of Laboratory Animals’ (2000) and ‘Safeguarding the welfare of Lab Animals’ (2001). Anyone working with lab animals must comply with these codes.
In addition, the Dierexperimentencommissie (DEC) of the UG has formulated internal guidelines to standardize University practices. These guidelines comprise the University’s opinions about the discomfort codes, the choice of species and ethical considerations.
Animal experiments: from application to execution
CCD
The Centrale Commissie Dierproeven (CCD) is a national committee which makes decisions to grant or reject project licenses for experiments based on the research applications. On its website, the CCD publishes non-technical summaries of the licenses it has granted.
NCad is another important national committee. NCad’s role is to bring about improvements in the application of the 3R principle and the ethical assessment thereof in scientific and applied research and in teaching activities, in order to minimize the use of laboratory animals both nationally and internationally.
DEC
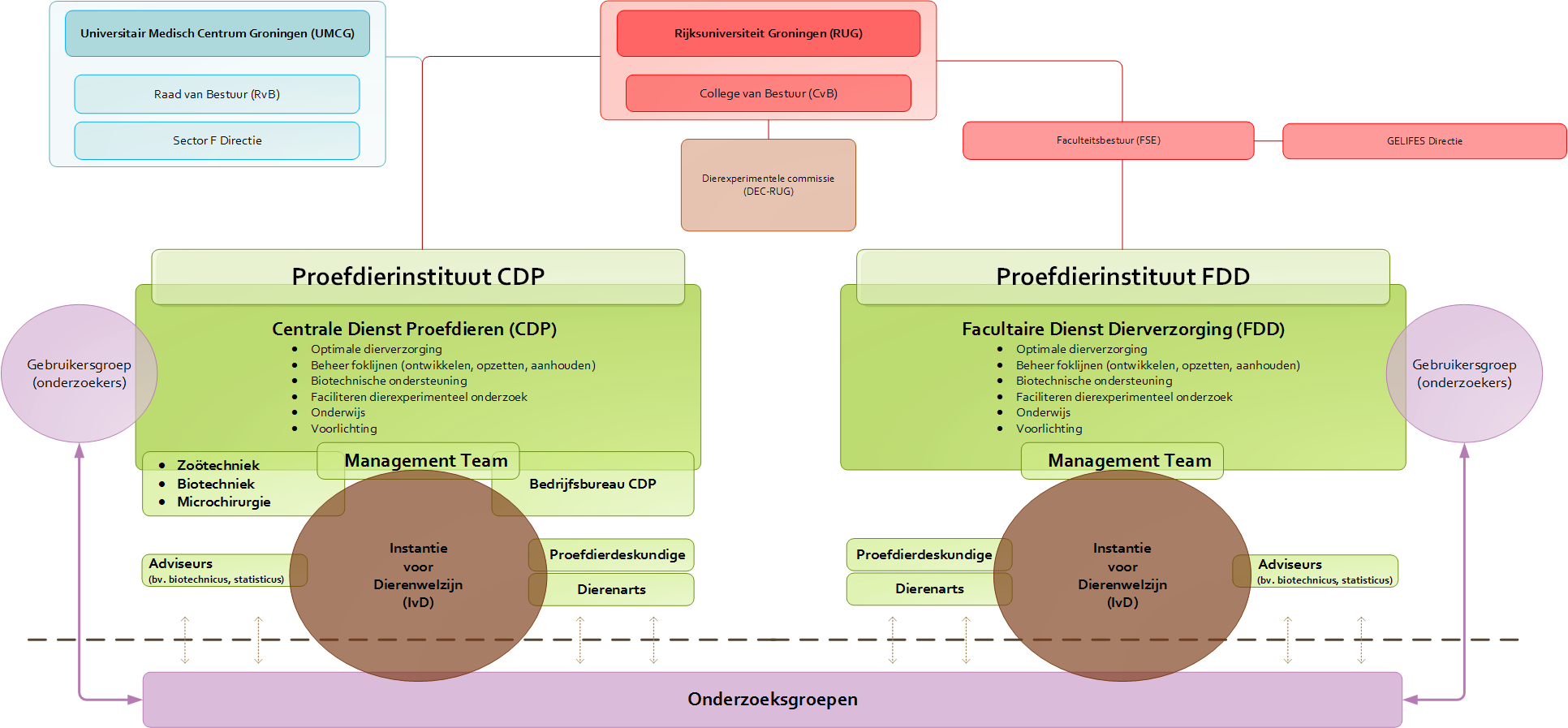
The UG has an impartial animal experiments committee (DEC–RUG) which assesses the use of lab animals under the auspices of the CCD, using the CCD’s opinions and guidelines. It also abides by generally applicable viewpoints from the various codes of practice. The DEC–RUGmembership includes experts in laboratory animals and their protection, animal experiments, alternatives for such experiments and ethical assessment.
The DEC assesses all research proposals in the light of current legislation and regulations. It also weighs the benefits of animal experiments against the discomfort caused to the animals to be used.
The intrinsic value of each animal is central to the decision whether an animal experiment is ethically acceptable or not. However, other considerations also play a role, for example an animal’s psychological complexity (cf. primates), the societal status of a species based on factors such as social closeness (cats and dogs), historical value (agricultural animals) and social relationship (seals).
The UG and the UMCG do not have facilities for experiments conducted with primates, and the UG has formulated a separate point of view on this issue (in Dutch only).
IvD
An important change in the revised Wod is that institutions must combine their expertise concerning animal welfare in an Instantie voor Dierenwelzijn (IvD). The IvD assesses the animal welfare aspects of a research project that has previously been approved by the DEC and the CCD and ensures that it can be properly implemented. It also advises researchers about the application of the 3R principle and supervises the research preparations and the skills and training of the researchers involved.
The IvD membership includes a designated veterinarian, the animal facility’s Location Supervisor, a scientist and, if necessary, an external expert such as a radiation specialist or biological safety officer.
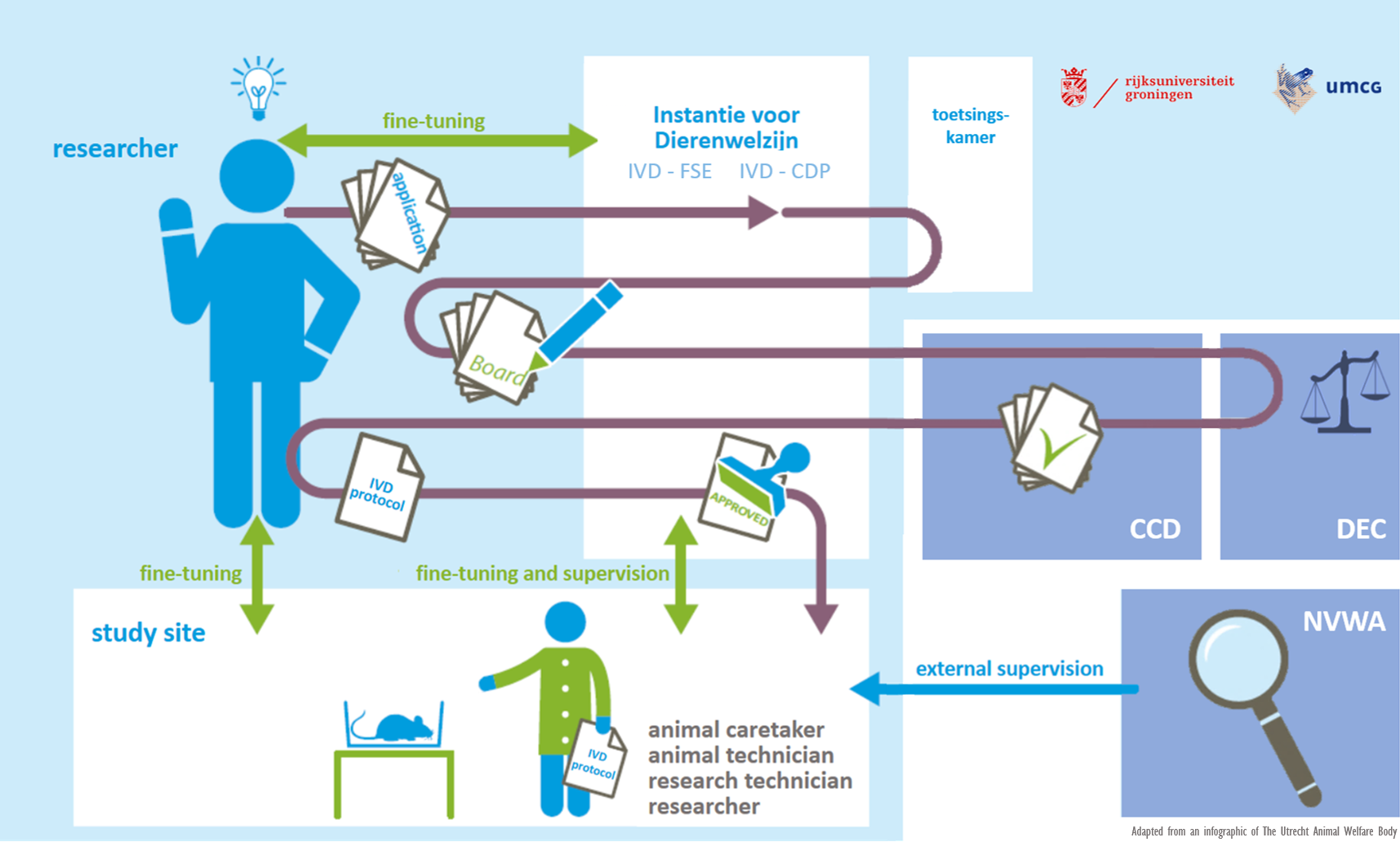
Article 14c of the Experiments on Animals Act lists the tasks of an Animal Welfare Body in five points (14c.1a to 1e). Article 14c.1c states that the IvD ‘guarantees the establishment and review of internal procedures concerning monitoring, reporting and follow-up with regard to the wellbeing of the animals housed in the institution’s animal housing facilities’. In other words, the Article states that the IvD organizes the laboratory animals’ guaranteed wellbeing and produces a record of it.
For each IvD protocol, the UG’s IvDs check whether the animal study will be carried out using animals that are caught in the wild. If so, the IvD checks whether the required flora and fauna dispensation has been obtained. This internal process will not change and continue to be used.
IvD platform
Both Animal Welfare Bodies (AWBs) at the University of Groningen are affiliated with the national AWB Platform. Members of the Groningen AWBs actively participate in various working groups, including the Antibiotics working group and the Breeding Coordinators working group. Additionally, one AWB member serves on the board of the AWB Platform. The AWB Platform is part of DALAS and represents more than 90% of all AWBs in the Netherlands.
In addition to monthly regular meetings, the AWB Platform maintains contact with various governmental organizations such as the CCD, NCad, LVVN, and the NVWA. The goal for the past year was to convene at least once annually with these organizations to discuss topics relevant to daily practice. This goal was achieved.
Besides serving as a discussion partner for governmental organizations, the AWB Platform also aims to facilitate the exchange of knowledge among the various Dutch AWBs.
There are eight active working groups within the AWB Platform: Experimental Design & Statistical Analysis, Breeding Coordinators, Peer Audit, Individual Housing, Article 9 Criteria, Antibiotics Policy, Severity Assessment, and the Registration Booklet. The output of these working groups is shared with the AWBs.
This year, in collaboration with NCad, the AWB Platform organized the “Harry Blom Consultation.” The aim of this event is to explore current dilemmas in research practice from various perspectives. The theme this time was “Refinement of test procedures, housing, and care of laboratory animals.” Several experts shared their research findings and insights. Most activities organized by the AWB Platform are attended by one or more AWB members from Groningen.
End of experiments
Adoption
According to the revised Dutch Animal Experimentation Act (Wet op de dierproeven), the Animal Welfare Body (IvD) provides advice on rehoming arrangements, including guidance on the appropriate socialisation of animals released for adoption. The license holder is of the opinion that it is not in the best interest of the laboratory animals or private individuals to adopt animals used in experiments. An exception is made for animals housed in semi-natural conditions at the University of Groningen (UG), such as birds and fish. These animals can be rehomed by private individuals. The license holder does not permit these animals to be traded.
In 2024, 51 zebra finches, 22 rock doves, and 1 red junglefowl were adopted. Additionally, 181 Java sparrows were returned to the breeder after use, and 2 parrotlets and 1 American kestrel were exchanged with the breeder because these individuals proved more difficult to train for the research. Finally, 140 three-spined sticklebacks were released into the wild.
Euthanasia
In most cases adoption is not possible, for example because the brain and/or other organs and body parts are required for further study and analysis. In that case, the animals will be euthanized at the end of the experiment. This is a step which neither animal carers nor researchers take lightly. The most common euthanasia procedure is one which the animals hardly notice. They are placed in a box containing a mixture of oxygen (O2) and carbon dioxide (CO2). Then the CO2 concentration is slowly increased, causing the animals to gradually lose consciousness and then pass away peacefully. Sometimes the nature of the experiment requires a different euthanasia procedure. In such cases, too, the method chosen must result in the least possible discomfort for the animal. In some cases, animals develop complications over the course of an experiment, which may lead them to suffer more than expected. Researchers will then apply the principle of the humane end point. They will remove the animal from the experiment when its suffering threatens to become unacceptable and then euthanize it to prevent further suffering.
Aims of animal experiments
By far the most animal experiments were carried out to help answer a scientific question. The figure below indicates what these questions entailed. In addition to answering scientific questions, animal experiments were also conducted within the scope of teaching and training, involving, for example, students and animal technicians.
Table Zodoende
| RuG 10500 | Fundamental research | Applied and translational research | Protection of animal species | Education | Breeding with discomfort not used | Total |
| Mice | 6,130 | 746 | 159 | 282 | 7,317 | |
| Rats | 730 | 80 | 205 | 1,015 | ||
| Guinea pigs | 15 | 14 | 29 | |||
| Other rodents | 39 | 39 | ||||
| Other mammals | 8 | 8 | ||||
| Other birds | 1,539 | 12 | 1,551 | |||
| Xenopus | 147 | 147 | ||||
| Zebrafish | 50 | 50 | ||||
| Seabass (temperate basses) | 17 | 17 | ||||
| Other fish | 304 | 76 | 380 | |||
| Total | 8,962 | 826 | 93 | 390 | 282 | 10,553 |
NB empty rows and columns are not shown.
The tabel does not include animals that have been terminated without prior procedures.
Discomfort
Lab animals will always experience some degree of discomfort. The revised Wod divides discomfort into four categories. Discomfort need not take the form of pain; stress and anxiety are also regarded as discomfort. The table below shows the degrees of discomfort and the percentages of animals involved in 2024.
%
Terminal / non-recovery
%
Mild discomfort
%
Moderate discomfort
%
Severe discomfort
%
Exceeding severe discomfort
Breeding efficiency
The UG and the UMCG breed animals themselves, particularly (transgenic) mice and rats. Not all bred animals end up in an experiment. We call these animals ‘surplus animals’ or ‘breeding surplus’.
A breeding surplus is unfortunately unavoidable. Animals in an experiment often have to be as identical as possible in order to obtain reliable research results. For example, they must be the same age and sex or born under identical circumstances. Also, not all animals possess the desired genetic characteristics. For an experiment involving 60 identical transgenic mice, for example, as many as 170 mice may have to be bred: read more about this on the website of the Stichting Informatie Dierproeven. In addition, a substantial part of the breeding is needed to maintain unique or valuable breeding lines.
Reducing the number of animals that are bred but not used in experiments remains a high priority for both the University of Groningen (UG) and the national government. In 2024, as in previous years, approximately one-fifth of all bred animals were used in experiments. This proportion is higher for non-transgenic lines, where all offspring are generally suitable for use. In contrast, transgenic lines often produce offspring with genotypes not suitable for experimental use.
The total number of bred animals (mice and rats) in 2024 was approximately 3,000 lower than in 2023. The absolute number of bred animals that were not used in experiments and were euthanised as surplus was about 5,000 fewer than in 2023, and 6,000 fewer than in 2022. This continued decrease is partly due to the use of cryopreservation for breeding lines that are not immediately needed. Experience has shown that most of these lines are not reactivated for breeding within a few years. This confirms the value of cryopreservation in preventing unnecessary breeding.
However, the decline in surplus breeding figures appears to have plateaued. The UG recognises that the number of unused animals remains high and continues to make efforts to reduce the number of surplus animals euthanised without being used.
The University of Groningen and the UMCG are at the forefront of increasing breeding efficiency through clear communication between researchers and animal caretakers (supply and demand) and reducing the breeding surplus by focusing on cryopreservation of breeding lines that are no longer actively used.

Cryopreservation
Cryopreservation is a technique in which eggs or sperm from a breeding line that is not needed for a longer period of time are frozen instead of keeping the line alive. When the breeding line is needed again, a fertilized egg is inserted into a pseudopregnant female. In the meantime, no animals are needed to maintain the line.
Since 2020, the UG has been offering to cryopreserve inactive breeding lines (lines that are no longer used for animal experiments).
In 2024, twenty mouse lines were discontinued, and eight new lines were initiated. On balance, fewer breeding animals were present for both mice and zebrafish.
In 2024, the UG also focused on developing the technical capacity to cryopreserve zebrafish lines, in order to avoid unnecessary maintenance of these lines as well. Overall, sustained efforts remain necessary to further reduce the percentage of unused animals in the coming years.
Replacement, Reduction, Refinement
The UG and the UMCG apply the 3R principle to research and teaching involving laboratory animals: replacement and reduction of the number of animals and refinement of the experiments in which they are used. Essentially, this means that we use as few animals as possible and conduct animal-free experiments whenever possible. Furthermore, we try to minimize the discomfort experienced by the animals. The Animal Welfare Committee (IvD) helps researchers to put these guidelines into practice.
Replacement
Researchers are only allowed to conduct an animal experiment if there are no other options. Where possible, we use alternatives to animal experiments in teaching and research, replacing laboratory animals with invertebrates, cells, tissues, computer simulations, video training or slaughterhouse material.
Reduction
Efforts must be made to reduce the number of animals required in each experiment through a research design specifying the minimum number of animals necessary to achieve reliable findings. This can be achieved, for example, by using standard strains so that the results are more comparable or by conducting a pilot study first.
Sometimes lab animals can be used again after the original experiment, in a follow-up or unrelated experiment or in a teaching activity. In 2023, 2.2% of animals was used again.
Refinement
Researchers, animal carers, animal technicians and designated veterinarians are always trying to refine all aspects of animal use and animal welfare. Optimum accommodation and adequate application of research techniques and anaesthesiology should minimize the animals discomfort. Social animals such as rats, for example, are kept in groups, which reduces their stress levels.
By refining animal experiments, we improve the animals’ welfare, which is not only good for them but benefits the quality of research too.
Applying the 3R principle in teaching activities involving lab animals
Training
The Centrale Dienst Proefdieren (CDP) applies the 3R principle as much as possible when lab animals are used for teaching purposes. When inexperienced students are first introduced to a technique, synthetic materials are used as much as possible. Students learning to suture, for example, first practice on a piece of chamois leather.
Microsurgical techniques are first practised using a piece of latex glove under the microscope and then on artificial vessels. If the students’ hand-eye coordination is sufficiently developed, they are allowed to continue with live rats. Synthetic materials are thus used whenever possible. Ultimately, however, the technique to be mastered must be practised in a live organism, since a living animal presents students with a system that is too complex to mimic with synthetic materials. To further reduce the number of animals used, instruction videos have been produced for all relevant biotechnical procedures covered during student training, so that no animals have to be used to demonstrate the techniques. Animals used in teaching are always anaesthetized before an invasive surgical procedure and are euthanized before the anaesthesia wears off, to prevent unnecessary discomfort. By thoroughly training the staff involved in animal experiments, the CDP aims to improve the quality of animal experiments and the animals’ welfare.
Anatomy practical
All Bachelor students of Biology take an anatomy and physiology practical involving the dissection of a rat. Until 2015, these rats were always euthanized shortly before the start of the practical and presented untreated to the students because this procedure results in the best specimens. In frozen and subsequently thawed specimens, certain essential structures proved difficult to see. Over the year, there are sufficient surplus animals available from breeding and invasive and non-invasive experiments to meet the needs of this practical. From the perspective of animal welfare, however, it is undesirable to keep these animals alive until the start of the practical. This is also uneconomical. For this reason, until 2015, the rats used were purchased from a commercial breeder. Despite the fact that almost all these animals were surplus animals from their breeding lines, this situation was less than ideal, not least because of the stress caused to the animals, for example during transport.
For these reasons, 2016 witnessed the start of a highly successful pilot in which surplus rats from our own breeding programme and experiments were embalmed. These embalmed rats proved highly useful in the practical because all the important structures were preserved well, which was not the case with the frozen specimens.
To embalm these rats, the Fix for Life method developed by Leiden University Medical Centre was used. This method employs an embalming fluid which is (virtually) free of the toxic and irritant substances such as formaldehyde and phenol that are commonly used to preserve tissues. This makes the method extremely suitable for teaching purposes. Another advantage is that the embalming fluid has a less offensive smell.
Embalming the rats has proven a win-win situation. First, rats no longer have to be purchased and transported, and our own surplus animals can now serve a useful purpose. At the same time, the specimens have proven extremely suitable for the practical, and their use is less taxing on students.
Organization and facilities
To guarantee optimum animal care and effective research, two modern animal experiment facilities have been set up: CDP at the UMCG and the Facultaire Dienst Dierverzorging (FDD) at the Linnaeusborg.
All animal studies at the UG and the UMCG are conducted either in nature or in one of the laboratories with special animal testing facilities. We take the utmost care to provide the best possible accommodations for lab animals, since they will live out almost their entire lives there. Providing accommodation thus involves more than simply meeting the statutory requirements. The CDP and the FDD have been completely renovated in 2009 and 2011, respectively, and are now among the most modern facilities in Europe. The temperature, lighting and atmospheric humidity in the animal quarters can be precisely controlled.
Inspections by the NVWA
In 2024 the NVWA conducted two visits: a three-day audit at the Linnaeusborg location and an inspection at the CDP location.

About the University of Groningen
The University of Groningen is a research university with a global outlook, deeply rooted in Groningen, City of Talent. The UG is in the top 100 of several important ranking lists. It is very popular with its 30,000 students and staff (5250 FTE) from the Netherlands and abroad, who are encouraged to make the most of their abilities. Talent is nurtured, and the keyword is quality. The University is committed to actively cooperating with its partners in society, with a special focus on its research themes Healthy Ageing, Energy and Sustainable Society.
 “We primarily look at the immune system in the lungs. We try to use as few laboratory animals as possible by using alternative models that can mimic certain aspects of lung diseases. However, the interaction with the immune system is quite complex because the immune system is not located in just one place in the body, but actually everywhere. This aspect is still missing in our alternative models. To properly test medication and understand the interaction between lung cells and immune cells, sometimes you need the whole system, and thus a laboratory animal.
“We primarily look at the immune system in the lungs. We try to use as few laboratory animals as possible by using alternative models that can mimic certain aspects of lung diseases. However, the interaction with the immune system is quite complex because the immune system is not located in just one place in the body, but actually everywhere. This aspect is still missing in our alternative models. To properly test medication and understand the interaction between lung cells and immune cells, sometimes you need the whole system, and thus a laboratory animal. “I research the type of learning that we humans use to learn our speech. This is called vocal learning. Children do this naturally, but when we learn a new language as adults, we also use this method. It is a special form of learning where we try to imitate the sounds and tones we hear. Not only humans do this, but songbirds also learn in this way. In humans, you can make brain scans, but you cannot conduct tests at the cellular level. Therefore, we use laboratory animals for this research.
“I research the type of learning that we humans use to learn our speech. This is called vocal learning. Children do this naturally, but when we learn a new language as adults, we also use this method. It is a special form of learning where we try to imitate the sounds and tones we hear. Not only humans do this, but songbirds also learn in this way. In humans, you can make brain scans, but you cannot conduct tests at the cellular level. Therefore, we use laboratory animals for this research. “I research the effects of radiation as used in radiotherapy to cure cancer. I mainly study the effects of radiation on normal tissue. What causes the side effects that a patient experiences when a tumor is irradiated? You can essentially eliminate a tumor with radiation, but that radiation also has to pass through healthy tissue surrounding the tumor. This healthy tissue can then be damaged. Healthy tissue thus determines the dose the patient can receive, and sometimes the dose cannot reach the level needed to eliminate the tumor. But even then, tissue damage can occur to healthy tissue, which can cause significant discomfort for the patient and, in rare cases, can be fatal.
“I research the effects of radiation as used in radiotherapy to cure cancer. I mainly study the effects of radiation on normal tissue. What causes the side effects that a patient experiences when a tumor is irradiated? You can essentially eliminate a tumor with radiation, but that radiation also has to pass through healthy tissue surrounding the tumor. This healthy tissue can then be damaged. Healthy tissue thus determines the dose the patient can receive, and sometimes the dose cannot reach the level needed to eliminate the tumor. But even then, tissue damage can occur to healthy tissue, which can cause significant discomfort for the patient and, in rare cases, can be fatal. “I am interested in the movement of animals. I mainly focus on flying, but in my field, we study how animals run, swim, and move. This is because we know very little about it. Understanding this has significant biological value. You can offer various solutions to the world just by understanding how nature works. That is my greatest interest. But it is also very important from the perspective of animal welfare. What is the behavior, and how can an animal move well? It is also very important for other areas within biology, such as ecology, evolutionary biology, or developmental biology. Movement is a fundamental aspect of how organisms live on Earth. It is how they gather food, find partners, reproduce, and migrate, so it is truly an essential biological requirement.
“I am interested in the movement of animals. I mainly focus on flying, but in my field, we study how animals run, swim, and move. This is because we know very little about it. Understanding this has significant biological value. You can offer various solutions to the world just by understanding how nature works. That is my greatest interest. But it is also very important from the perspective of animal welfare. What is the behavior, and how can an animal move well? It is also very important for other areas within biology, such as ecology, evolutionary biology, or developmental biology. Movement is a fundamental aspect of how organisms live on Earth. It is how they gather food, find partners, reproduce, and migrate, so it is truly an essential biological requirement. “We are researching the use of antidepressants during pregnancy and how it affects offspring. We want to see if the brains of newborns develop differently, as we know that the behavior is different. We would like to investigate the underlying causes of this. Research in pregnant women is, of course, not ethically possible, and we don’t want to do that either, because we would have to treat healthy pregnant women with antidepressants. There is research in humans, where studies are conducted at young and later ages, but that doesn’t progress quickly. If you want to look at the underlying mechanisms in the body, you really need the brain and have to specifically study it. So that simply can’t be done in humans. It is also important to mention that when testing new medications, it is still mandatory to first test the safety of the medication in laboratory animals. This is especially important when it comes to the next generation.
“We are researching the use of antidepressants during pregnancy and how it affects offspring. We want to see if the brains of newborns develop differently, as we know that the behavior is different. We would like to investigate the underlying causes of this. Research in pregnant women is, of course, not ethically possible, and we don’t want to do that either, because we would have to treat healthy pregnant women with antidepressants. There is research in humans, where studies are conducted at young and later ages, but that doesn’t progress quickly. If you want to look at the underlying mechanisms in the body, you really need the brain and have to specifically study it. So that simply can’t be done in humans. It is also important to mention that when testing new medications, it is still mandatory to first test the safety of the medication in laboratory animals. This is especially important when it comes to the next generation.





